 
In chemistry, it is crucial to accurately measure their quantities. To confirm your answer, compare the calculated value to the weighted mass displayed on the periodic table. What is molar mass All substances are made up of atoms or molecules. There are nearly 118 elements (at present) of which nearly 94 occur naturally on Earth. Compound are substances which are formed by two or more different types of elements that are united chemically in fixed proportions. The atomic mass of oxygen (use percent natural abundance data from Table 2.5.1) would be calculated as follows:Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + (%3)(mass 3) Elements are pure substances which are composed of only one type of atom. For chlorine, there are only two naturally occurring isotopes so there are only two terms.Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + ⋯Īnother example: oxygen exists as a mixture that is 99.759% 16O, 0.037% 17O and 0.204% 18O. This is repeated until there is a term for each isotope. The weighted average is determined by multiplying the percent of natural abundance by the actual mass of the isotope. For normal samples from earth with typical isotope composition, the atomic weight can be approximated by the standard atomic weight or the conventional atomic weight. Weve talked about it in other videos, you could view this 12. The given mass of K (4.7 g) is a bit more than one-tenth the molar mass (39.10 g), so a reasonable ballpark estimate of the number of moles would be slightly greater than 0.1 mol. The molar mass of atoms of an element is given by the relative atomic mass of the element multiplied by the molar mass constant, M u 1 × 10 3 kgmol 1. So if we first look at carbon, carbon, we see from this periodic table of elements, has a molar mass of 12.01 grams per mole. The atomic mass of an element is the weighted average of the atomic masses of the naturally occurring isotopes of that element. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. Why? The reason is that we need to take into account the natural abundance percentages of each isotope in order to calculate what is called the weighted average. 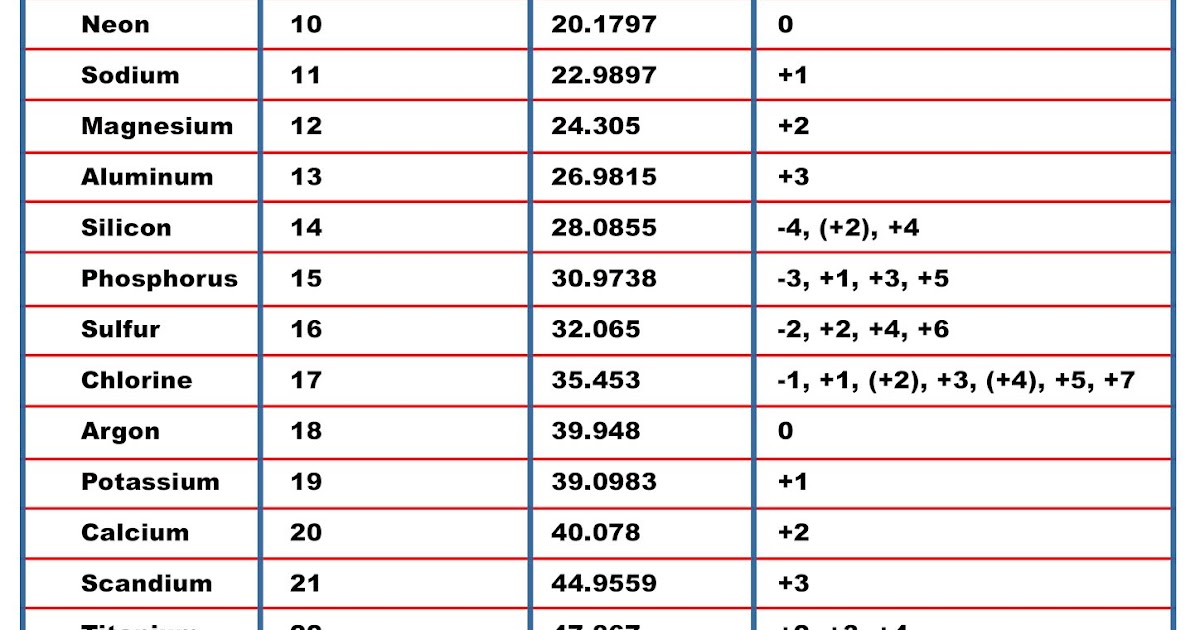
The PDF is suitable for high-resolution color printing for desk or wall-chart display. = 35.968 \,u \nonumber \]Īs you can see, the average atomic mass given in the last column of the table above (35.453) is significantly lower. The periodic table contains NIST’s latest critically evaluated data for atomic properties of the elements. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
